BIOMECHANICS
Designing, testing and evaluating new treatment modalities to advance the quality of patient care and outcomes.
History:
The Legacy Biomechanics Laboratory was established in 1999 by Michael Bottlang, Ph.D, and Steven Madey, M.D. It serves as a resource for basic and applied research with the goal to design, test and evaluate new treatment modalities that will advance the quality of patient care and outcomes. Biomechanical engineering provides an important link between the knowledge base of expert scientists and clinicians and the development of new products by industry.
Research Focus:
The laboratory focuses on classical biomechanical research concerns to improve the treatment of orthopedic injuries, and the prevention of traumatic injuries. As such, it relies on close collaboration between biomechanical engineers and orthopedic surgeons.Collaborations with the Level 1 Trauma Center at Legacy Emanuel Medical Center and with Legacy's orthopedics department have produced compelling findings that have led to the development of new devices and processes which addressed unmet needs.
Reimagining the Bicycle Helmet |
|
 |
The Legacy Biomechanics Laboratory has recently conducted groundbreaking research on the cause of brain injury in falls, highlighting the need for mitigating of ROTATIONAL forces during impact.
The laboratory aided in testing of a novel bicycle helmet technology, termed “WaveCel”, that proves vastly improved protection from brain injury. Today, close to 1 million WaveCel helmets have been sold, and the WaveCel helmet was featured in the latest “Bicycling Magazine: 100 Best Products". This successful translation of research into a consumer product highlights the community outreach and global impact in a consumer product. |
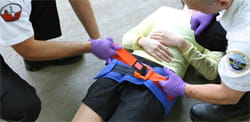
Pelvic Sling |
|
|
We developed a stabilization device for life-threatening pelvic fractures. Our invention, marketed as “SAM Sling”, represents the de facto standard for emergent stabilization of pelvic ring fractures in the US, and it is sold in 48 countries (>50,000 units / year). Dr. Madey, a Legacy Orthopaedic Surgeon, has been the key collaborator in this project. The SAM Sling is produced locally (Sam Medical) in Wilsonville. |
Rib Plate System |
|

|
We developed an implant system for rib fracture fixation that has induced a paradigm shift towards surgical fixation of chest wall injuries, which has been shown to significantly decrease morbidity, mortality, and treatment cost. Our system has been licensed and is sold under the name MatrixRIB in 26 countries (>2,500 procedures/ year). Dr. William Long, the former Director of Legacy’s Level I Trauma Center, was a key collaborating surgeon, and this project highlights our close collaboration with Legacy Trauma Services. Legacy Research technology saves woman’s life |
Funding:
Funding for projects of Dr. Bottlang’s research program has been solicited from the National Institute of Health: helmet research for brain injury prevention is funded by the National Institute for Neurological Disorders and Stroke (NINDS); research on bone fracture treatment and tissue engineering was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases; and research on pelvic stabilization has been funded by the Department of Defense (DoD), and the US Office of Naval Research. In addition, funding has been provided by Legacy Health's foundations.

